- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
Very Early Isavuconazole Toxicity in Allogeneic Stem Cell Transplant Recipient
- Home
- Back to Journal
- Article Details
Abstract
We report a case of 67-year-old male with acute myeloid leukemia, who developed graft-versus-host disease and started isavuconazole for possible invasive aspergillosis. Six days later he developed severe anorexia and asthenia. Blood level of isavuconazole from day 3 of treatment was >10mg/L. Antifungal therapy was stopped and after 7 days the patient gradually improved. Isavuconazole toxicity should be suspected in case of side effects, even after short treatment duration and confirmed by blood level testing.
Keywords: Isavuconazole, Invasive aspergillosis, Blood level, Toxicity, Side effect
Introduction
Treatment of invasive fungal infections remains a challenge particularly among immunocompromised patients such as hematopoietic Stem Cell Transplant (SCT) recipients or those with haematological malignancies [1]. Triazole antifungal agents are first-line agents for the prevention and treatment of these infections. Voriconazole is recommended as primary treatment for Invasive Aspergillosis (IA) and posaconazole is primarly indicated as salvage therapy for patients with IA or prophylaxis in neutropenic patients [2]. The class of azole antifungals is characterized by a number of major shortcomings, including drug-drug interactions and nonlinear and saturable pharmacokinetics with need for therapeutic drug monitoring, particularly in case of voriconazole [3]. Additionally, typical side effects of azoles, include hepatic and gastrointestinal toxicity (liver enzymes increase, nausea, vomiting,) and QTc prolongation [3–5]. While the relationship between the exposure to voriconazole and posaconazole and the efficacy has been established, the association between blood levels and toxicity is mainly known for voriconazole and its neurological and visual side effects, and to the lesser extent, hepatic toxicity [6–8]. Isavuconazole is the newest broad-spectrum triazole approved in 2015 for treatment of Invasive Aspergillosis (IA) and mucormycosis in adult patients, with significantly lower rate of side effects, particularly liver toxicity compared to voriconazole [9].
Recently we reported that isavuconazole was efficacious and well tolerated, with an increase in the rate of side effects, mainly gastrointestinal, associated with prolonged administration and high serum levels [10].
Here we present a case of 67 year-old male who discontinued isavuconazole therapy after few days due to progressive anorexia and severe asthenia and in whom very high serum levels were detected at day 3 after treatment onset.
Case
We present a case of 67 year-old male with acute myeloid leukaemia diagnosed in February 2018, who in August 2018 was in second complete remission and underwent allogeneic SCT from a haploidentical donor with high dose post-transplant cyclophosphamide as Graft versus Host Disease (GvHD) prophylaxis.
The patient developed chronic GvHD and was initially successfully treated with corticosteroids (1mg/kg of methylprednisolone) in February 2019. In April 2019 he was admitted for chronic GvHD flare with lichenoid oral lesions and rash, diarrhoea, dysphagia, weight loss of 4 kg and progressive pancytopenia with neutropenia (400 neutrophils/ml). Additionally, he reported new onset of productive cough. An increase in C-Reactive Protein (CRP) was noted (119 mg/L, upper limit of 5 mg/L), TC demonstrated lung lesions compatible with bacterial pneumonia, and pharyngeal swab confirmed colonisation with P. aeruginosa resistant to ceftazidime and piperacillin/tazobactam. Serum galactomannan and beta-D-glucan were negative. Antibiotic therapy with meropenem and linezolid was started for bacterial pneumonia, caspofungin was started because of oral, and possibly, oesophageal candidiasis and acyclovir and trimetoprim/sulfametoxazole prophylaxis were continued. Immunosuppressive treatment with methylprednisolone (1.5mg/kg initially) was started and cyclosporin dose was increased, with rapid improvement of rash, diarrhoea, oral lesions and neutropenia.
Twenty days after admission, and 5 days after discontinuing antibiotic treatment, the patient developed a new episode of productive cough and repeated haemoptysis. Cavitated lung lesions were noted on lung CT and pharyngeal swab grew a Multi-Drug Resistant (MDR) P. aeruginosa. The diagnosis of MDR P. aeruginosa pneumonia and possible IA was made. Intravenous treatment with isavuconazole (standard loading dose of 200mg tid for 6 doses, and then 200mg daily), ceftolozane/tazobactam (1.5g tid) and amikacin (500mg/day) was started awaiting the results of broncoalveolar-lavage. The remaining concomitant medications included pantoprazole 40mg bid, trimetoprim/sulfametoxazole 960 mg thrice weekly, valacyclovir 1000mg/day, methylprednisolone 40mg/day, furosemide 20mg/day, cyclosporine 60mg/day iv, total parenteral nutrition 1500ml/day with 16 IU of rapid insulin and 20mEq of magnesium sulphate. Chronic renal insufficiency was present with creatinine clearance of 47 ml/min. BMI was 16, MELD score was 9.
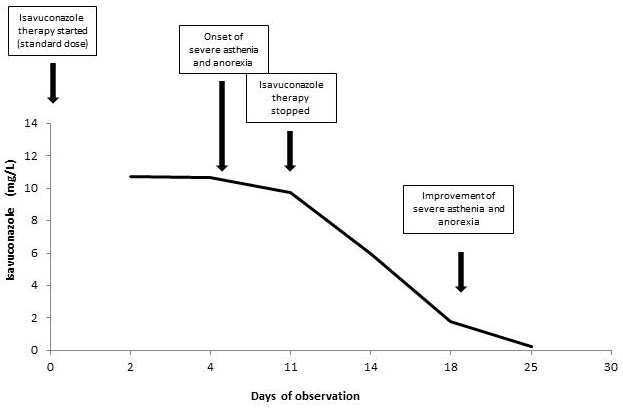
After 6 days from starting isavuconazole, amikacin and ceftolozane/tazobactam the patient developed progressive anorexia and severe asthenia and was unable to walk, with no evident neurological deficit. There was no new increase in liver enzymes or cholestasis indexes except for bilirubin (ALT 38U/L, ALP 108U/L, GGT 83U/L, bilirubin 1.5mg/dL), no worsening of coagulation parameters (AP 60, INR 1.3) and GvHD was under control (no rash, no diarrhoea, no neutropenia, no new oral lesions). Since the symptoms continued worsening and the patient stopped almost any daily activity and food intake, isavuconazole therapy was discontinued after 10 days of therapy, while amikacin and ceftolozane/tazobactam were continued. Isavuconazole blood levels, assessed by an HPLC-UV method as previously described [10], were 10.7 mg/L on the 3rd day of isavuconazole administration and 9.8 mg/L on the first day after stopping isavuconazole. The patient gradually improved starting from day 7 after the discontinuation of isavuconazole. Isavuconazole blood levels are shown in figure 1.
Figure 1: Isavuconazole blood levels during the period of observation.
Although diagnostic workup for IA was negative, the patient was considered at high risk of IA due to GvHD and its treatment. Thus, sixteen days after discontinuing isavuconazole, posaconazole prophylaxis with tablet formulation was started at low dose and without loading dose (400 mg/daily). After 2 and 4 days posaconazole blood levels were, respectively, 1.47 mg/L and 2.56 mg/L, but on day 5 of treatment an increase in ALT was recorded (ALT 230 U/L and GGT 245 U/L, compared to, respectively, 52 U/L and 90 U/L before posaconazole treatment), and posaconazole therapy was discontinued.
Discussion
This report suggests that isavuconazole toxicity should be suspected in patients developing side effects, even in case of very short treatment duration and could be confirmed by blood level testing. Although cohort studies did not find association between isavuconazole blood levels and toxicity [11], it is probable that high levels are more frequently associated with side effects, as reported for other azoles [6, 8]. Indeed, a 31% and 32% incidence of neurotoxicity were found at trough concentration of voriconazole > 5.5 mg/L and > 5 mg/L, respectively [6, 12]. Denning et al. reported abnormal liver function or liver failure in 6/22 patients with trough concentration > 6 mg/L [13]. Other adverse events attributable to voriconazole high levels included nausea, headache, abnormal taste and asthenia [13], while the most common adverse events associated with posaconazole are gastrointestinal, such as nausea and vomiting [14]. Additionally, weakness has been reported as a rare side effect of voriconazole and posaconazole, and asthenia was reported for voriconazole as <1% of adverse events (2/623) [13], or patients (1/194) [15], both during short (< 6 months) and long term treatment (respectively, 2/319 and 2/109), while anorexia was described in 3/319 and 5/109 patients during, respectively, short and long term treatment [16]. Although fatigue has been reported in 10.5% of patients in the isavuconazole trials (vs. 6.9% in the voriconazole arm) [9], we report here a case of severe fatigue which onset and resolution has close time association with high isavuconazole blood levels.
Recently, we reported good tolerability of isavuconazole, with adverse events, mainly gastrointestinal and anorexia, occurring in six patients (31.6%) who received prolonged isavuconazole treatment and had high blood levels [10]. However, none of them had such high isavuconazole blood levels in the first 10 days of treatment (median, 3.7 mg/L, range 0.64-7.3).On the contrary, other authors reported rapid onset of isavuconazole toxicity in a SCT patients who developed fatal hepatic veno-occlusive disease after 5 days from starting isavuconazole, with blood levels of 5 and 8 mg/L on days 4 and 8 of treatment, even though other concomitant causes might have been present [17].
Our patient did not develop liver toxicity despite high blood levels, confirming what already known that isavuconazole has liver metabolism but liver toxicity has been reported as generally mild, rarely requiring discontinuation [9, 18]. On the contrary, he developed rapid liver toxicity after posaconazole administration, with normal blood levels and despite the lack of loading dose and reduced daily dose, which might be indicative of impaired hepatic metabolism of azoles and high blood levels. Our hypothesis is that such a reduced metabolism of azoles, and consequent toxicity, was caused by malnutrition and decreased liver function due to GvHD, together with being underweight (BMI 16) rather than by a genetic mechanism, since a year before, our patient received posaconazole tablets at standard dose for prophylaxis for 17 days during induction chemotherapy, with normal blood levels (median value 2.27, range: 1.6–3.6) and no toxicity.
In conclusion, this case report highlights a rare possibility of rapid onset of isavuconazole toxicity associated with high blood levels, possible in patients with low BMI and poor hepatic metabolism. Identification of factors associated with early high blood levels warrants further studies.
Conflict of Interest
M. has received speaker and advisory board fees from Gilead, P?zer, Janssen and MSD. All other authors: none to declare.
References
- D. Neofytos, D. Horn, E. Anaissie, W. Steinbach, A. Olyaei, et al. (2009) Epidemiology and Outcome of Invasive Fungal Infection in Adult Hematopoietic Stem Cell Transplant Recipients: Analysis of Multicenter Prospective Antifungal Therapy (PATH) Alliance Registry. Clin. Infect. Dis. 48: 265–273. [View]
- C. Lass-Flörl (2011) Triazole antifungal agents in invasive fungal infections: A comparative review, Drugs. 71: 2405–2419 [View]
- D. Allen, D. Wilson, R. Drew, J. Perfect (2015) Azole antifungals: 35 years of invasive fungal infection management. Expert Rev Anti Infect Ther 13: 787-798. [View]
- Pfizer, (2008). VFEND ® Tablets, Packag. Inser. [View]
- I. Astellas (2018) Pharma US, Cresemba (isavuconazole) [package insert], Northbrook, IL. [View]
- A. Pascual, T. Calandra, S. Bolay, T. Buclin, J. Bille, O. Marchetti (2008) Voriconazole Therapeutic Drug Monitoring in Patients with Invasive Mycoses Improves Efficacy and Safety Outcomes. Clin. Infect. Dis 46: 201–21. [View].
- P.F. Troke, H.P. Hockey, W.W. Hope (2011) Observational study of the clinical efficacy of voriconazole and its relationship to plasma concentrations in patients. Antimicrob Agents Chemother 64: e00483-20. [View]
- H.R. Ashbee, R.A. Barnes, E.M. Johnson, M.D. Richardson, R. Gorton, W.W. Hope (2014) Therapeutic drug monitoring (TDM) of antifungal agents: Guidelines from the british society for medical mycology. J. Antimicrob. Chemother 69: 1162–1176 [View]
- J.A. Maertens, I.I. Raad, K.A. Marr, T.F. Patterson, D.P. Kontoyiannis, et al. (2016) Isavuconazole versus voriconazole for primary treatment of invasive mould disease caused by Aspergillus and other filamentous fungi (SECURE): A phase 3, randomised-controlled, non-inferiority trial. Lancet 387: 760-769. [View]
- E. Furfaro, A. Signori, C. Di Grazia, A. Dominietto, A.M. Raiola, et al. (2019) Serial monitoring of isavuconazole blood levels during prolonged antifungal therapy. J. Antimicrob. Chemother 74: 2341–2346. [View]
- A. V. Desai, L.L. Kovanda, W.W. Hope, D. Andes, J.W. Mouton, et al. (2017) Bonate, Exposure-response relationships for isavuconazole in patients with invasive aspergillosis and other filamentous fungi. Antimicrob Agents Chemother 61: 01034-17. [View]
- M.J. Dolton, J.E. Ray, S.C.A. Chen, K. Ng, L.G. Pont, A.J. McLachlan (2012) Multicenter study of voriconazole pharmacokinetics and therapeutic drug monitoring. Antimicrob. Agents Chemother 56: 4793– 4799. [View]
- D.W. Denning, P. Ribaud, N. Milpied, D. Caillot, R. Herbrecht, et al. (2002) Efficacy and Safety of Voriconazole in the Treatment of Acute Invasive Aspergillosis, Clin Infect Dis 34: 563–571. [View]
- W. D., B. K., T. M., S. G., (2010) Efficacy and safety of standard long-term posaconazole antifungal prophylaxis in allogeneic stem-cell transplant recipients. Clin Microbiol Infect 17: 507–515[View]
- R. Herbrecht, D.W. Denning, T.F. Patterson, J.E. Bennett, R.E. Greene, et al. (2002) Voriconazole versus amphotericin B for primary therapy of invasive aspergillosis. N Engl J Med 347:408-415. [View]
- I.I. Raad, J.R. Graybill, A.B. Bustamante, O.A. Cornely, V. Gaona-Flores (2006) Safety of Long-Term Oral Posaconazole Use in the Treatment of Refractory Invasive Fungal Infections. Clin Infect Dis 42: 1726–1734. [View]
- A. Mesini, G. Cangemi, E. Palmisani, C. Dufour, E. Castagnola (2018) Hepatic veno-occlusive disease during isavuconazole administration. J Chemother 30: 63-64. [View]
- I. Kyriakidis, A. Tragiannidis, S. Munchen, A.H. Groll (2017) Clinical hepatotoxicity associated with antifungal agents. Expert Opin. Drug Saf 16: 149-165. [View]
Article Type
Case Report
Publication history
Received: August 12, 2024
Accepted: August 22, 2024
Published: August 25, 2024
Citation:
Furfaro E, Raiola AM, Di Grazia C, Varaldo R, Angelucci E, Mikulska M (2024) Very Early Isavuconazole Toxicity in Allogeneic Stem Cell Transplant Recipient. Clar J Infect Dis Ther 01(02): 42–45.
E. Furfaro1, A.M. Raiola2, C. Di Grazia2, R. Varaldo2, E. Angelucci2 and M. Mikulska1,3*
1Division of Infectious Diseases, Department of Health Sciences (DISSAL), University of Genoa, Genoa, Italy
2Division of Hematology and Bone Marrow Transplantation, IRCCS Ospedale Policlinico San Martino, Genoa, Italy
3Division of Infectious Diseases, IRCCS Ospedale Policlinico San Martino, Genoa, Italy
*Corresponding author
Malgorzata Mikulska, MD PhD,
Division of Infectious Diseases,
Department of Health Sciences (DISSAL),
University of Genoa,
Italy and IRCCS Ospedale Policlinico San Martino,
Genova, Italy,
Largo Rosanna Benzi, 10, 16132,
Genova, Italy;
Phone: +39 010 555 4649;
Fax: +39 010 555 6712;