- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
Insecticide Susceptibility of Aedes aegypti (Diptera: Culicidae) from Cabo Verde
- Home
- Back to Journal
- Article Details
Abstract
Aedes aegypti mosquitoes are major vector of arboviruses such as dengue virus, yellow fever virus, chikungunya and Zika virus. In Cabo Verde two epidemic diseases (dengue and Zika) were already recorded and Ae. aegypti was identified as the only vector. The vector control program has been using deltamethrin 0.05% in indoor spraying for controlling adult’s population since 1999, and 1% granulated temephos for larvae. In this study we evaluate the resistance status of four insecticides in Ae. aegypti populations from eight municipalities, comprising three islands.
Mosquitoes were sampled using wood stick pallets to collect eggs on the field population and reared to adults in the laboratory to perform the WHO insecticide susceptibility test protocol. Females mosquitos from 2 to 5 days old were tested to determine Ae. aegypti insecticide susceptibility status. The tested populations showed resistance to carbamate (bendiocarb 0,1% and propoxur 0,1%) and were susceptible to fenitrothion 1%. Only one population showed resistance to deltamethrin 0,05%, out of eight tested populations. Fenitrothion is a good option for controlling Aedes aegypti population in Cabo Verde, in the event of insecticide rotation.
Key words: Aedes aegypti, adults, insecticide, resistance, Cabo Verde.
Introduction
Aedes aegypti is a major vector of arboviruses such as Dengue Virus (DENV 1–4), Yellow Fever Virus (YFV), Chikungunya (CHIKV) and Zika Virus (ZIKV) [1–3]. Currently, these are the most important arboviruses concerning public health, accountable to emergent and re-emergent epidemics and outbreaks worldwide [4, 5].
In Cabo Verde two epidemics have took place, Dengue in 2009 and Zika in 2015 [6–9]. The mosquito vector identified to date is Aedes aegypti, found to be present in all the inhabited islands [10–12].
There are no effective vaccines available to these diseases, except to yellow fever, thus the main way to control the disease transmission is vector control. The insecticide application is an effective way to interrupt disease transmission in vector control programs, but the sustainability of insecticide use is threatened by two major limitation. First is the development of resistance to insecticides in mosquito’s population, and the second is the limited number of new insecticides available [13–16].
For more than twenty years the vector control strategies in Cabo Verde are based on indoor residual spray with deltamethrin 0.05% for adults, and 1% granulated temephos for larvae [17]. Regarding temephos, Aedes aegypti larvae from Santiago island was tolerant to this insecticide in 2012 and 2014 [18].
A study conducted in 2009 demonstrated that Aedes aegypti from Cabo Verde was susceptible to deltamethrin 0.05%, fenotrothion 1%, lambda-cyhalothrin 0.05%, permethrin 0.75, with possible resistance to propoxur 0.1%, but resistant to DDT4% (dichlorodiphenyltrichloroethane) [19]. In 2012, Rocha and collaborators (2015) showed for the first time insecticide resistance to deltamethrin (3µg/µL) and cypermethrin (8 µg/µL), but susceptibility to malathion (30 µg/µL) [18].
This study aimed to evaluate the susceptibility of Aedes aegypti mosquitoes to the insecticides used in Cabo Verde in the context of public health, since the last study of this kind was in 2012. Due to the prolonged use of same insecticide, this work will provide crucial information allowing the vector control program to implement more effective and efficient measures/methods including an insecticide rotation strategy for the control of the adult forms of Aedes aegypti, preventing their abundance and higher risk for disease transmission.
Methods
Study Area
CaboVerde is a volcanic archipelago, located about 550 km from the West African coast, with a total area of 4 033Km2. It consists of ten islands (one uninhabited) divided into two groups of islands (Barlavento and Sotavento) administratively comprising 22 municipalities with a total resident population of 537661 inhabitants. It has hot and dry, arid to semi-arid climate with an annual average temperature around 25ºC, low rainfall, where two seasons are evident: the dry season, from December to June and the rainy season from August to October [20]. The study took place from September 2018 to December 2019 on three islands (Maio, Santiago and Fogo) in Sotavento.
Eggs Sampling and Larvae Rearing
To collect Aedes aegypti eggs, Fay & Eliason (1966) adapted ovitraps (OVT traps) were placed inside and outside the dwellings [21]. The traps remained on site for 5 days, and then collected and transported to the Medical Entomology Laboratory (LEM) at the National Institute of Public Health (INSP).
The palettes made of wood sticks with eggs were left for 24 hours to dry, with temperature ranging from 26–28ºC and a relative humidity 70–80%. The eggs were counted and stored for maximum of 2 weeks and then placed in vat with water for hatching and maintenance of the larvae. The larvae were fed with grinded fish food (tropical mix flakes) to adult stage. Adults emerged from larvae in the insectary were fed with a 10% sucrose solution.
Test Procedures
Tests were carried out on unfed female Aedes aegypti mosquitoes with 2 to 5 days old, according to WHO tube susceptibility test protocol [22]. Batches of 20 – 25 mosquitos were exposed to four different insecticides: 0.05% deltamethrin (pyrethroid class), 1% fenitrothion (organophosphate class), 0.1% bendiocarb and 0.1% propoxur (carbamate classe).
For each test, a control containing up to 25 mosquitoes was used, subject to the same procedures, except for exposure to the insecticide. Knockdown effect in each exposure tube was recorded in 10, 15, 20, 30, 40, 50 and 60 minutes. After exposure, the mosquitoes were transferred to the resting tubes placed in an upright position with 10% sucrose in moistened cotton.
Mosquitoes that remained alive after the tests were killed by freezing at –20ºC for 20 minutes, counted and stored at –20ºC. All specimens were identified using the dichotomous key according to Ribeiro, Ramos, Capela and Pires (1980) [22].
Data Analysis
The mortality rate was recorded 24 hours post mosquitoes exposure to insecticides and in control group. Whenever mortality rate in control was greater than 5% the Abbot formula was used to correct mortality, as following [29]
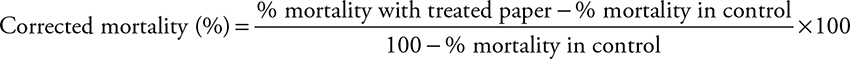
Resistance status was analysed according to the WHO (2016a) criteria, in which mortality rate under 90% shows resistance, 90% – 97% suggest possible resistance to confirmation and ≥98% indicates susceptible population.
All the data from the assay was registered and analyzed in Excel sheet (Microsoft Office 2013).
Results
A total of 2 226 female Aedes aegypti mosquitoes were tested to four different insecticides and 600 females’ mosquitoes served as controls during testing. Four insecticides (0.05% deltamethrin, 1% fenitrothion, 0.1% bendiocarb and 0.1% propoxur) were tested in four municipalities - Praia, São Lourenço dos Órgãos, São Salvador do Mundo and Tarrafal. Due to the low density on mosquito’s population during the period of sampling, we could not perform the test in all municipalities. One insecticide (0.05% deltamethrin) was tested in the municipalities of Ribeira Grande, Santa Catarina, Maio and São Filipe.
The lower mortality was recorded to deltamethrin 0.05% iin Tarrafal, 88%, suggesting resistance to this insecticide. In Praia and São Lourenço dos Órgãos was registered 91% and 92%, respectively, which suggest possible resistance to be confirmed to deltamethrin. Mosquitoes from the four municipalities (Praia, São Lourenço, São Salvador do Mundo and Tarrafal de Santiago) were susceptible to fenitrothion 1%, showing mortality equal or greater than 98%. Resistance to bendiocarb 0.1% and propoxur 0.1% was confirmed in those four municipalities, even though in São Salvador do Mundo and Tarrafal bendiocarb 0.1% was possible resistant to be confirmed (Table 1).
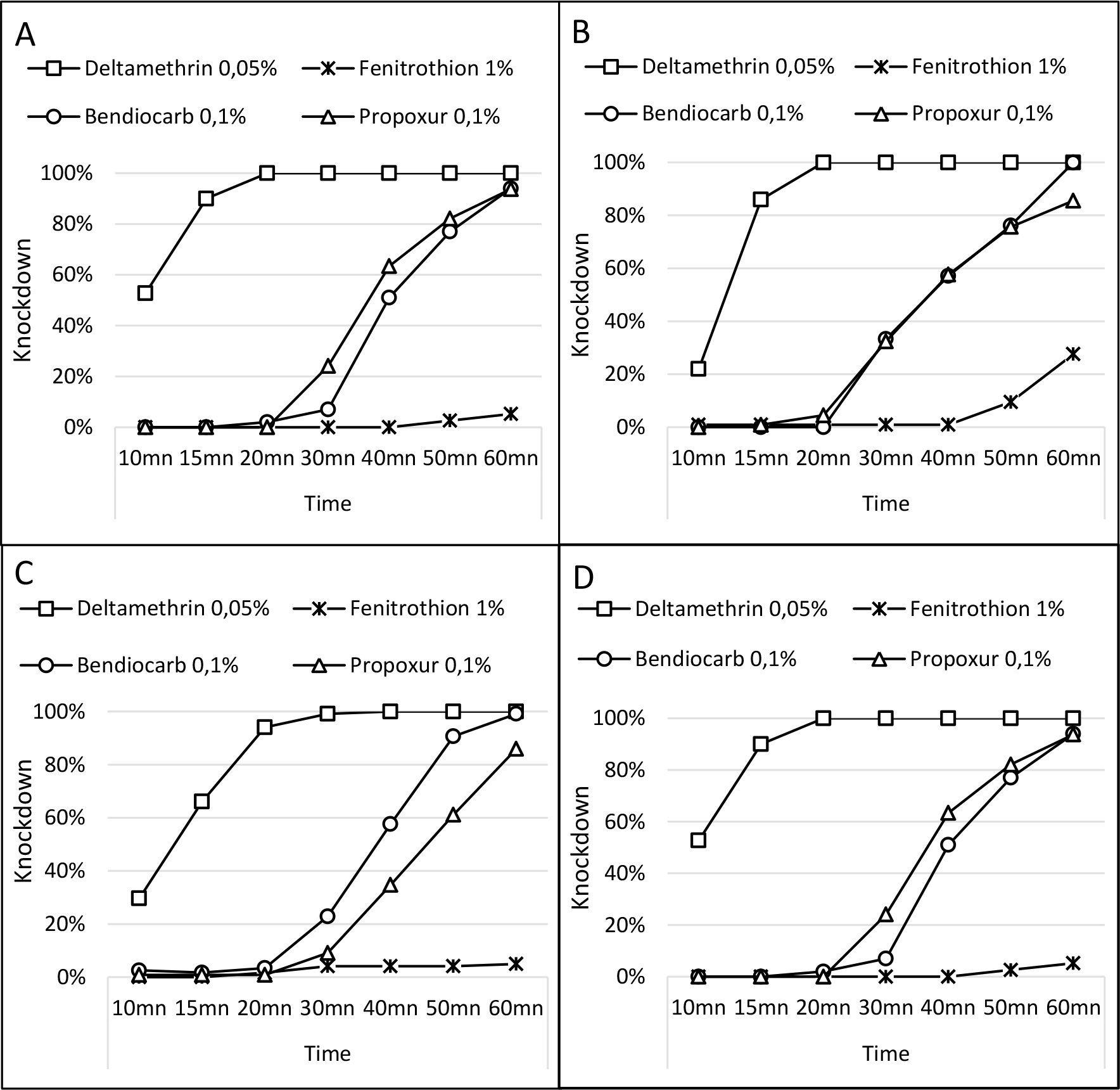
Knockdown pattern
In the Praia municipality, the 1-hour exposure to deltamethrin 0.05% showed that at 20 minutes more than 90% of mosquitos knocked-down. For bendicarb 0.1% and propoxur 0.1% the KdT90 was at 60min. Only 5% of mosquitoes was knocked down after 1-hour exposure to fenotrothion 1% (Figure 1A). In the municipality of São Lourenço dos Órgãos the KdT90 was registered at 20 min for deltamethrin 0.05% and bendiocarb 0.1% and 100% of mosquitoes was knocked after 1-hours exposure to both insecticides. No KdT90 was recorded for propoxur 0.1% and fenitrothion 1% since the knockdown rate was 86% and 28%, respectively, after 1-hour exposure (Figure 1B). The KdT90 was obtained after 20 min and 50 min of exposure to deltamethrin 0.05% and bendiocarb 0.1%, respectively, in the municipality of São Salvador do Mundo. No KdT90 was recorded for propoxur 0.1% and fenotrothion 1% as the 1-hour exposure showed the overall knockdown rate of 86% to the first and 5% to the last insecticide (Figure 1C). In Tarrafal the KdT90 for deltamethrin 0.05% was recorded 15min after exposure, and in 20 min all mosquitoes were knocked down. Both bendiocarb 0.1% and propoxur 0.1% showed 94% of knocked down mosquitoes in 60min exposure. Only 5% of mosquitoes were knocked down after 1-hour exposure to fenotrothion 1% (Figure 1D).
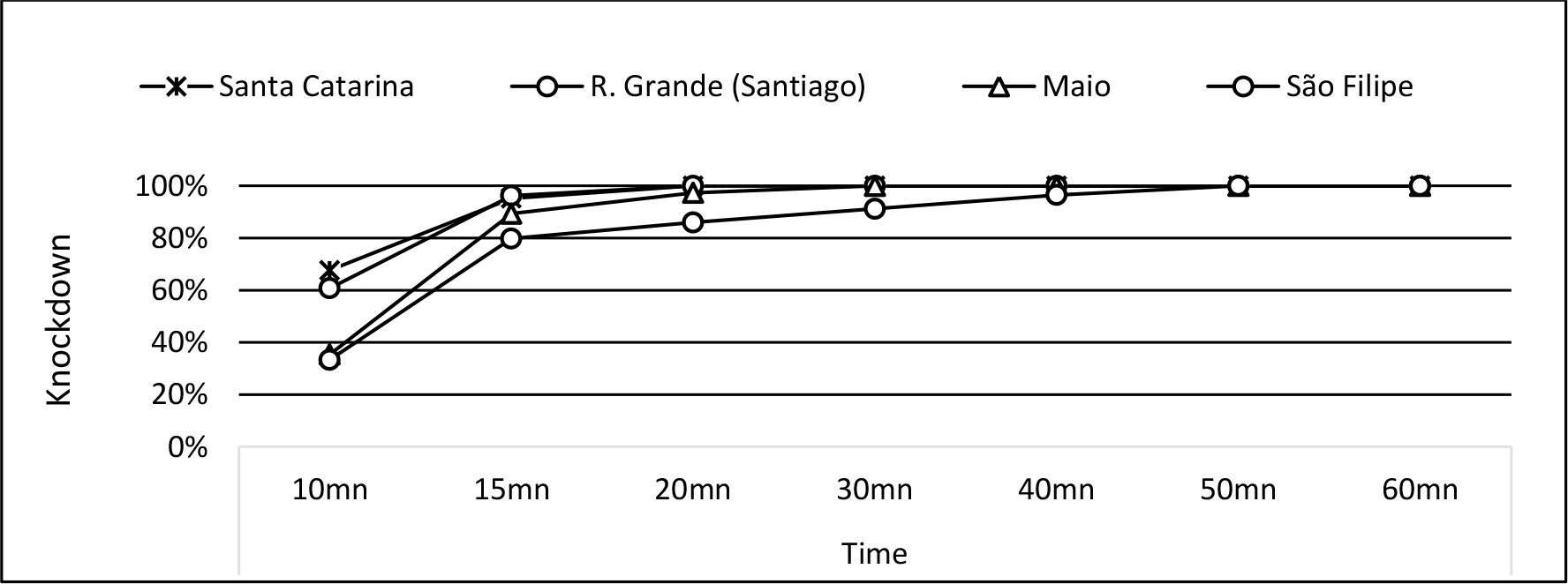
In the municipalities where only deltamethrin 0.05% was tested, after 1-hour exposure all mosquitoes were knockdown. The KdT90 was recorded 15min in Ribeira Grande and Santa Catarina, in Maio and São Filipe it was recorded in 20min and 30min after exposure, respectively (Figure 2).
Table 1: Mortality rate on field population of Aedes aegypti in the studied sites and the concentrations of insecticides tested.
|
Islands |
Municipalities |
Deltamethrin 0.05% |
Fenitrothion 1% |
Bendiocarb 0,1% |
Propoxur 0,1% |
||||||||||||
|
(pyrethroid) |
(organophosphate) |
(carbamate) |
(carbamate) |
||||||||||||||
|
N |
NC |
MR |
RS |
N |
NC |
MR |
RS |
N |
NC |
MR |
RS |
N |
NC |
MR |
RS |
||
|
Santiago |
Praia |
102 |
25 |
91% |
PRC |
111 |
25 |
100% |
S |
119 |
25 |
65% |
R |
119 |
25 |
64% |
R |
|
S. S. do Mundo |
118 |
25 |
100% |
S |
121 |
25 |
99% |
S |
118 |
25 |
98% |
S |
121 |
25 |
83% |
R |
|
|
Tarrafal |
110 |
25 |
88% |
R |
115 |
25 |
100% |
S |
100 |
25 |
95% |
PRC |
112 |
25 |
82% |
R |
|
|
S. L. dos Órgãos |
100 |
25 |
92% |
PRC |
105 |
25 |
99% |
S |
105 |
25 |
85% |
R |
111 |
25 |
82% |
R |
|
|
R. Grande |
107 |
25 |
93% |
PRC |
*** |
** |
** |
** |
** |
** |
** |
** |
** |
** |
** |
** |
|
|
Santa Catarina |
105 |
25 |
94% |
PRC |
*** |
** |
** |
** |
** |
** |
** |
** |
** |
** |
** |
** |
|
|
Maio |
Maio |
113 |
25 |
100% |
S |
*** |
** |
** |
** |
** |
** |
** |
** |
** |
** |
** |
** |
|
Fogo |
São Filipe |
114 |
25 |
99% |
S |
*** |
** |
** |
** |
** |
** |
** |
** |
** |
** |
** |
** |
N-number of mosquitoes; NC-number of mosquitoes used in control; MR-mortality rate; RS; Resistance status; R- resistant population; S-susceptible population; PRC- possible resistance to confirmation.
Figure 1: Knockdown curve pattern at 1 hour of exposure for the four insecticide concentrations tested. A – municipality of Praia, B – municipality of São Lourenço dos Órgãos, C – municipality of São Salvador do Mundo, D – municipality of Tarrafal.
Discussion
Insecticide resistance study in population of Aedes aegypti from Cabo Verde have shown resistance to three insecticides class. [18, 19]. This study shows resistance to pyrethroid and carbamate whilst the results from Dia and collaborators (2012) showed resistance to organochlorine. Organophosphate is the sole class where no resistance has been reported in populations of Aedes aegypti from Cabo Verde.
Aedes aegypti population from Tarrafal was resistant to deltamethrin, and population from Praia was susceptible according to our results. In Praia, probably there are greater insecticide selection pressure in mosquito’s population due to frequent use of insecticide for vector control [8, 23, 24]. In 2009, Aedes aegypti was susceptible to deltamethrin, fenitrothion, lambda-cyhalothrin and permethrin in Praia, nevertheless, in 2012 populations from Santiago island showed resistance to deltamethrin and cypermethrin [18, 19].
Figure 2: Knockdown curve pattern for the insecticide deltamethrin 0.05% in Ribeira Grande de Santiago, Santa Catarina de Santiago, Maio and São Filipe.
The main mechanism of resistance is related to a gene in the voltage-gated sodium channels of insects, known as knockdown resistance (Kdr), which is associated with resistance to organochlorines and pyrethroids. However, this mutation was not detected in Aedes aegypti population in 2012 [18]. Therefore, the phenotypic resistance described in 2012 could be associated with enzymatic detoxification mechanism, due to pressure in insecticide use mainly after the 2009 dengue epidemic [8].
Among the insecticides used in this study, only one study describes the concentration used for Aedes aegypti and many of them are tentative tests [15, 25]. Mosquitoes from different municipalities showed similar Knockdown curve pattern when exposed to the same insecticides and all the tested population were susceptible to fenitrothion and resistant to propoxur.
The pyrethroid’s insecticides concentrations used in the control of Aedes aegypti populations are less of those used in Anopheles control [15, 25]. Generally, vector control programs use the described insecticide concentration defined for Anopheles mosquitoes in malaria control programs, exposing local Aedes aegypti populations to higher concentration of insecticides and accelerating/ potentiation the development of resistance to insecticides. Populations of Aedes aegypti resistant to organochlorine (especially DDT) are widely recorded, and for pyrethroids (especially deltamethrin) and carbamates (especially propuxur) insecticide resistance is widely present in South America and Southeast Asia. Regarding the organophosphates malathion and fenitrothion, resistance in Aedes aegypti populations has only been recorded inPortugal, Madeira Region and Sudan [26, 27].
Conclusions
Regarding deltamethrin 0.05%, three population were susceptible, four population were resistant possible to be confirmed and one population was resistant out of height tested population.manipulation and preparation of insecticide, the actual insecticide pressure on the mosquitoes’ population and the frequent use towards others vectors are some features that could explain the patterns in the resistance status in Cabo Verde. Bendiocarb and propoxur are not suitable for Aedes aegypti population control, in Cabo Verde. In the possibility of insecticide rotation fenitrothion would be a good option for controlling Aedes aegypti population in Cabo Verde.
Acknowledgments
We acknowledge the National Institute of Public Health of Cabo Verde for supporting the study, and the National Program for vector control to all the Health facilities, health delegates, control vector agents, drivers, and administrators that provided their support during the entomological survey.
References
- Jentes ES, Poumerol G, Gershman MD, Hill DR, Lemarchand J, Lewis RF, et al. (2011). The revised global yellow fever risk map and recommendations for vaccination, 2010: consensus of the Informal WHO Working Group on Geographic Risk for Yellow Fever. The Lancet Infectious Diseases 11: 622–632. [View]
- Mayer SV, Tesh RB, and Vasilakis N (2017) The emergence of arthropod-borne viral diseases: A global prospective on dengue, chikungunya and zika fevers. Acta Tropica, 166: 155–163. [View]
- Gould E, Pettersson J, Higgs S, Charrel R, and de Lamballerie X (2017) Emerging arboviruses: why today? One Health 4: 1–13. [View]
- Mukhopadhyay S, Kuhn RJ, and Rossmann MG (2005) A structural perspective of the flavivirus life cycle. Nature Reviews Microbiology 3: 13. [View]
- Lopes N, Nozawa C, and Linhares REC (2014) Características gerais e epidemiologia dos arbovírus emergentes no Brasil. Revista Pan-Amazônica de Saúde 5: 55–64. [View]
- https://www.who.int/csr/don/2009_11_18/en/
- De Estudos G (2010) RELATÓRIO ESTATÍSTICO de 2009, 1–120.
- Associação Paranaense dos Criadores de Bovinos da raça Holandesa (ABCBRH) 2016, Relatório Estatístico 2015 Annu. Rep., p. 52.
- Lourenço J, de Lourdes Monteiro M, Valdez T, Rodrigues JM, Pybus O, and Faria NR (2017) Zika virus outbreak in Cabo Verde Islands, West Africa: early epidemiological findings. bioRxiv, 198952. [View]
- Alves J, Gomes B, Rodrigues R, Silva J, Arez AP, Pinto J, and Sousa CA (2010). Mosquito fauna on the Cape Verde Islands (West Africa): an update on species distribution and a new finding. Journal of Vector Ecology 35: 307–312. [View]
- Alves J, Pina AD, Diallo M, Dia I (2014). First report of Culex (Culex) tritaeniorhynchus Giles, 1901 (Diptera: Culicidae) in the Cabo Verde Islands. Zool. Caboverdiana 5: 4–19.
- Leal SDV, Fernandes Varela IB, Lopes Gonçalves AAL, Sousa Monteiro DD, Ramos de Sousa CM, Lima Mendonça MDL, and Osório HC (2020) Abundance and Updated Distribution of Aedes aegypti (Diptera: Culicidae) in Cabo Verde Archipelago: A Neglected Threat to Public Health. International Journal of Environmental Research and Public Health 17: 1291. [View]
- Brogdon WG, and McAllister JC (1998) Insecticide resistance and vector control. Emerging Infectious Diseases 4: 605. [View]
- Vontas J, Kioulos E, Pavlidi N, Morou E, Della Torre A, and Ranson H (2012) Insecticide resistance in the major dengue vectors Aedes albopictus and Aedes aegypti. Pesticide Biochemistry and Physiology 104: 126–131. [View]
- World Health Organization (2016a). Monitoring and managing insecticide resistance in Aedes mosquito populations—Interim guidance for entomologists.
- Smith LB, Kasai S, and Scott JG (2016) Pyrethroid resistance in Aedes aegypti and Aedes albopictus: Important mosquito vectors of human diseases. Pesticide Biochemistry and Physiology 133: 1–12. [View]
- Duarte EH, Correia EE, Varela CE, and Varela A (2012) Reproduction of mosquitoes (Diptera: Culicidae) in Santa Cruz, Santiago island, Cape Verde Islands. Zool Caboverdiana, 3: 29–36. [View]
- Rocha HDR, Paiva MHS, Silva NM, de Araújo AP, Camacho D, dos R, da R, de A, da Moura AJF, Gómez LF, Ayres CFJ, and de Melo Santos MAV (2015) Susceptibility profile of Aedes aegypti from Santiago Island, Cabo Verde, to insecticides. ActaTropica 152: 66–73. [View]
- Dia I, Diagne CT, Ba Y, Diallo D, Konate L, and Diallo M (2012) Insecticide susceptibility of Aedes aegypti populations from Senegal and Cape Verde Archipelago. Parasites & vectors, 5: 238. [View]
- Neves DJD, Silva V. de PR, Almeida RSR, Sousa F. de A. S. de and Da Silva BB (2017) General aspects of the climate in the Cabo verde archipelago. Ambiência, 13.
- Fay RW, and Eliason DA (1966) A preferred oviposition site as a surveillance method for Aedes aegypti. Mosquito News 26: 531–5. [View]
- World Health Organization (2016b). Test procedures for insecticide resistance monitoring in malaria vector mosquitoes.
- Depina AJ, Namountougou M, Leal SV, Varela IBF, and Monteiro DDS (2018b) Susceptibility of Anopheles gambiae Sl to the Insecticides in Praia, Cape Verde: A Country in the Pre-Elimination of Malaria. Vector Biol J 3: 2. [View]
- De Pina AJ, Andrade AJB, Dia AK, Moreira A, Faye O, and Seck I (2018a). Achievement of malaria pre-elimination in Cape Verde according to the data collected from 2010 to 2016. Malaria journal 17: 236. [View]
- World Health Organization (2016c). Discriminating Concentrations of Insecticides for Adult Mosquitoes. 1h exposure – WHO/CDS/CPC/MAL/98.12 and WHO Technical Report Series 818. Disponível em: https: //www.who.int/neglected_diseases/vector_ecology/resources/en/Diagnostic_concentrations.pdf; acesso em dezembro de 2019.
- Kamgang B, Yougang AP, Tchoupo M, Riveron JM, and Wondji C (2017) Temporal distribution and insecticide resistance profile of two major arbovirus vectors Aedes aegypti and Aedes albopictus in Yaoundé, the capital city of Cameroon. Parasites Vectors 10: 1–9. [View]
- Weetman D, Kamgang B, Badolo A, Moyes CL, Shearer FM, Coulibaly M, and McCall PJ (2018) Aedes mosquitoes and Aedes-borne arboviruses in Africa: current and future threats. International journal of environmental research and public health 15: 220. [View]
Article Type
Research Article
Publication history
Received: January 25, 2025
Accepted: February 19, 2025
Published: March 02, 2025
Citation:
Fernandes Varela IB, Sousa Monteiro DD, Lopes Gonçalves AA, Ramos de Sousa CM et al. (2025) Insecticide Susceptibility of Aedes aegypti (Diptera: Culicidae) from Cabo Verde. Clar J Infect Dis Ther 06(01): 306–313.
Isaias Baptista Fernandes Varela1*, Davidson Daniel Sousa Monteiro1, Aderitow Augusto Lopes Gonçalves1, Celivianne Marisia Ramos de Sousa1, Maria da Luz Lima Mendonça1, António Lima Moreira2, Adilson José De Pina3, Hugo Costa Osório4,5 and Silvânia Da Veiga Leal1
1Laboratório de Entomologia Médica, Instituto Nacional de Saúde Pública, Largo do Desastre da Assistência, Chã de Areia, Praia 719, Cabo Verde
2Programa Nacional de Luta contra o Paludismo, Ministério da Saúde e da Segurança Social, Cabo Verde
3Programa de Pré-Eliminação do Paludismo, CCS-SIDA, Ministério da Saúde e da Segurança Social, Várzea, Praia 855, Cabo Verde
4Instituto Nacional de Saúde Doutor Ricardo Jorge, Centro de Estudos de Vectores e Doenças Infecciosas, Avenida da Liberdade 5, 2965-575 Águas de Moura, Portugal
5Instituto de Saúde Ambiental, Faculdade de Medicina da Universidade de Lisboa, Av. Prof. Egas Moniz, Ed. Egas Moniz, Piso 0, Ala C, 1649-028 Lisboa, Portugal
*Corresponding author
Fernandes Varela IB,
Laboratório de Entomologia Médica,
Instituto Nacional de Saúde Pública,
Largo do Desastre da Assistência,
Chã de Areia,
Praia 719, Cabo Verde;