- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
Clinical Considerations on the Potential Interaction between SARS-Cov-2 and Hemoglobin
- Home
- Back to Journal
- Article Details
Keywords: COVID19; Oxygenation; Acute Respiratory Failure; ARDS
SARS-CoV-2 is a new virus that appeared in China at the end of last year [1], and has recently caused a pandemic outbreak worldwide. The infection of SARS-CoV-2 causes CoVid-19 disease and a significant portion of patients develop lung involvement that combines elements of pneumonia, inflammatory cytokine storm and alterations in the pulmonary circulation [2, 3]. All these abnormalities can lead to hypoxemia, which is sometimes very severe, and even to the development of an Acute Respiratory Distress Syndrome (ARDS) [4]. The diagnosis and management of these processes implies the periodic obtaining of arterial blood gases to adapt oxygen supplementation to the needs of the patient. In the most severe cases, the use of invasive or non-invasive ventilator support may also be necessary. Partial pressure of oxygen in arterial blood (PaO2) is the variable commonly used in clinics to assess the level of hypoxemia, as well as the index that relates this parameter to the inspiratory fraction of oxygen received by the patient (PaO2/FIO2), currently known as PaFi [5]. This was described approximately half a century ago and allows the grading of the severity of the disease in ARDS patients receiving oxygen supplements, although it has also been used in assessing the level of hypoxemia in other entities [6, 7]. However, the use of PaO2 as the main oxygenation index is based on the fact that its relationship with the oxygen saturation of hemoglobin (SaO2) is quite predictable under normal circumstances. This relationship takes the form of an italic ‘S’ and shows that in the intermediate part of the curve even small decreases in PaO2 have important consequences in SaO2, which in turn conditions a deficiency of oxygen supply to the tissues. This is due to the fact that approximately 95% of oxygen circulates bound to hemoglobin [8], and only a very small portion does so dissolved in plasma. Although the relationship between PaO2 and SaO2 may vary somewhat in clinical circumstances, depending on factors such as the partial pressure of CO2 in arterial blood (PaCO2), acid-base balance or temperature, it is generally accepted that they are minor changes with relatively little impact on patient status and management.
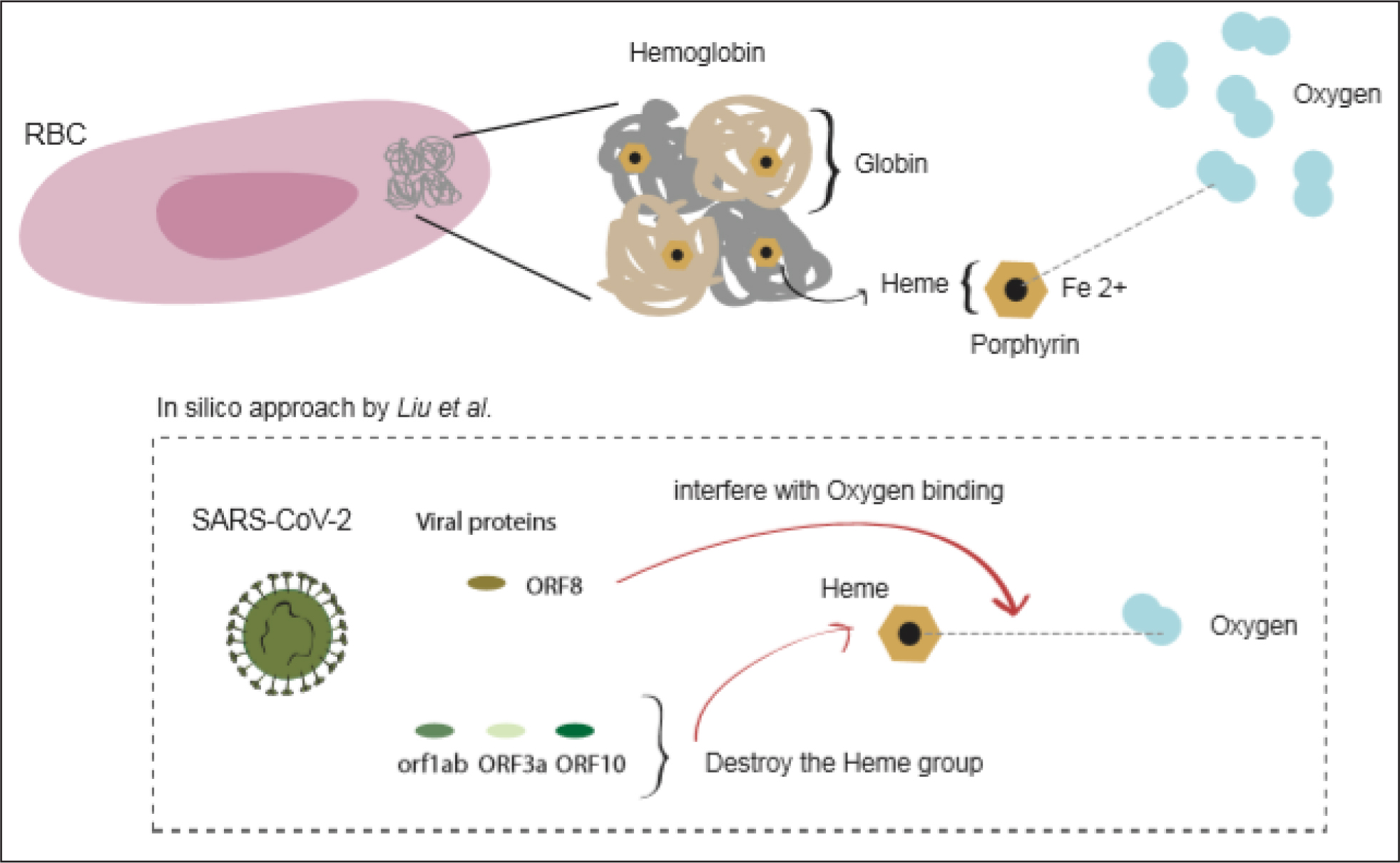
As previously mentioned, the main oxygen carrier in blood is hemoglobin, a complex molecule of 54 kD formed by four subunits or groups called ‘heme’, consisting of a porphyrin with an iron atom (Fe), attached to a globin. The Fe ion in particular is the hemoglobin element capable of reversibly binding to oxygen.
A very recent and controversial study using in silico methodologies has shown that certain proteins of the SARS-CoV-2 virus, such as ORF8, would be able to interfere with the Fe to oxygen binding (figure 1) [9]. Moreover, other viral proteins (orf1ab, ORF3a and ORF10) could directly destroy the heme group, and therefore hemoglobin, by releasing the Fe ion, with similar pathophysiological consequences [9]. These results would tally with the fact that many CoVid-19 patients show a reduced amount of hemoglobin in their blood [10], a finding that is associated with an increase in serum ferritin (perhaps to buffer Fe ions released from destroyed hemoglobin) [11], and inflammatory markers [12].
Figure 1: Graphical representation of the interactions between viral proteins and the hemoglobin molecule (see the text). Red Blood Cell.
From viral and immunological levels, iron is crucial for viral replication and many viruses can directly disrupt iron homeostasis [13]. However, whether this holds true also for SARS-CoV-2 needs experimental evidence. Moreover, on the one hand macrophages play a pivotal role in the regulation of iron homeostasis by preventing the release of hemoglobin from red blood cells. On the other, an excess of heme can act as an alarmin to activate the innate immune system and induce a pro-inflammatory polarization in macrophages [14, 15]. It is not yet known whether these cells can get productively infected with SARS-CoV-2, but they have been suggested as drivers for the cytokine storm observed in COVID-19 patients [16]. High ferritin levels together with the cytokine profiles observed in severe COVID-19 patients correlate with Haemophagocytic Lympho-Histiocytosis (HLH), a macrophage activation syndrome that leads to the dysregulation of the macrophage function with an excessive immune activation and tissue damage. The potential involvement of some SARS-CoV-2 proteins on disrupting iron metabolism and causing an excessive immune response in the case of COVID-19 needs to be taken with caution but raises interesting questions that exceed the clinical setting.
If Liu et al.’s in silico approach is confirmed in vivo it could have important consequences in the management of CoVid-19 patients. Under these circumstances, PaO2 would no longer be a good indicator of the patient’s oxygenation status, as we already know that it occurs for instance in carbon monoxide poisoning, and, as a logical consequence neither would PaFi be appropriate for this purpose. It would be more coherent to use the real value of SaO2 and its derivative, the SaO2/FiO2 index (or SaFi) as oxygenation markers. More controversial would be the use of saturation measured with the oximeter (or SpO2) for this same purpose [17, 18], since this instrument only gives an approximation to the real SaO2, which is based on the absorption of light through relatively translucent areas of the patients’ body. Fortunately, most blood gas analyzers are today equipped with cooximetry, which allows obtaining the true value of SaO2. Therefore, the calculations, sometimes erroneous, that were previously used to calculate the approximate value of SaO2 (through Kelman subroutine or the Severinghaus nomogram)8 have been excluded in most care centers.
In conclusion, until further data on the effects of SARS-CoV-2 on the hemoglobin molecule (an essential oxygen transporter to maintain aerobic metabolism) is available, caution should be taken with the weight that clinicians give to PaO2 and PaFi in patients with CoVid-19. This is especially true if there is an obvious discrepancy between the values of measured PaO2 and measured SaO2, since the former could lead to underestimating the severity of the patient’s actual blood and tissue oxygenation status.
References
- Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y et al. (2020) Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395: 497–506. [View]
- Menter T, Haslbauer JD, Nienhold R, Savic S, Hopfer H, Deigendesch N et al. (2020) Post-mortem examination of COVID19 patients reveals diffuse alveolar damage with severe capillary congestion and variegated findings of lungs and other organs suggesting vascular dysfunction. Histopathology (in press). [View]
- Boraschi P (2020) COVID-19 Pulmonary involvement: Is really an interstitial pneumonia? Acad Radiol 2020 (in press). [View]
- Wu C, Chen X, Cai Y, Xia J, Zhou X, Xu S et al. (2020) Risk factors associated with Acute Respiratory Distress Syndrome and death in patients with Coronavirus disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med (in press). [View]
- Kirby RR, Downs JB, Civetta JM, Modell JH, Dannemiller FJ, Klein EF et al. (1975) High level Positive End Expiratory Pressure (PEEP) in acute respiratory insufficiency. Chest 67:156–63. [View]
- Yan LP, Liu ZB, Wu M, Ge YP, Zhang Q (2020) Effect of lncRNA MALAT1 expression on survival status of elderly patients with severe pneumonia. Eur Rev Med Pharmacol Sci 24: 3959–64. [View]
- Kim N, Lee SH, Joe Y, Kim T, Shin H, Oh YJ (2020) Effects of inhaled iloprost on lung mechanics and myocardial function during one-lung ventilation in Chronic Obstructive Pulmonary Disease patients combined with poor lung oxygenation. Anesth Analg 130: 1407–14. [View]
- John Burnard West, Andrew Luks (2016) West’s Respiratory Physiology: The Essentials. JB West & AM LUks (eds.). Tenth Edición. Wolters Kluwert Health. Philadelphia 2015. [View]
- Liu W, Li H (2020) COVID-19: Attacks the 1-beta chain of hemoglobin and captures the porphyrin to inhibit human heme metabolism ChemRxiv Preprint. [View]
- Lippi G, Mattiuzzi C. Hemoglobin value may be decreased in patients with severe coronavirus disease 2019. Hemat Transfus Cell Ther (in press). [View]
- Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z et al. (2020) Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 395: 1054–62. [View]
- Ye Q, Wang B, Mao J (2008) Cytokine Storm in COVID-19 and treatment. J Infect (in press). [View]
- Drakesmith H, Prentice A (2008) Viral infection and iron metabolism. Nat Rev Microbiol 6: 541–52. [View]
- Soares MP, Hamza I (2016) Macrophages and Iron Metabolism. Immunity 44: 492â€504. [View]
- Soares MP, Bozza MT (2016) Red alert: labile heme is an alarmin. Curr Opin Immunol 38: 94–100. [View]
- Merad M, Martin JC (2020) Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nature Rev Immunol (in press). [View]
- Paulsen SM, Killyon GW, Barillo DJ (2002) High-frequency percussive ventilation as a salvage modality in adult respiratory distress syndrome: a preliminary study. Am Surg 68: 852–6. [View]
- Chen W, Janz DR, Shaver CM, Bernard GR, Bastarache JA, Ware LB (2015) Clinical characteristics and outcomes are similar in ARDS diagnosed by oxygen saturation/Fio2 ratio compared with Pao2/Fio2 ratio. Chest 148: 1477–83. [View]
Article Type
Short Commentary
Publication history
Received: May 19, 2020
Accepted: May 26, 2020
Published: May 28, 2020
Citation:
Gea J, Gea-Mallorquí E (2020) Clinical Considerations on the Potential Interaction between SARS-Cov-2 and Hemoglobin. Med Case Rep Ther Stud 01(02): 15–17.
Joaquim Gea1* and Ester Gea-Mallorquí2
1Respiratory Department, Hospital del Mar – IMIM. DCEXS, Universitat Pompe
2Viral Immunology, Nuffield Department of Medicine, University of Oxford, Oxford, UK
*Corresponding author
Joaquim Gea,
Respiratory Department,
Hospital del Mar,
Pg Marítim 27,
08003 Barcelona,
Spain;